NGL prices have been rising fast since the middle of this year, but the same cannot be said for the price of natural gas. So how does this market scenario play out for gas processors who make their money extracting NGLs from gas? It plays out pretty darn good. In Part 1 of this series, we looked at how the relationship between the price of NGLs versus natural gas can be assessed by the Frac Spread, and concluded that things are definitely looking up for gas processing economics. But we also concluded that the Frac Spread misses the impact of a few key factors, including the BTU value and composition of the inlet gas stream. So today we’ll see what it takes to incorporate those factors into our assessment and, in the process, do a deep dive into the math of gas processing to examine the relationship between volumetric capacity, gallons of NGLs per 1,000 cubic feet of natural gas (GPMs) and moles. Today, we continue our latest expedition into the wilds of gas processing.
In Part 1, we reviewed the calculation methodology, the history and the pros and cons of the Frac Spread — the difference between the price of natural gas and the weighted average price of NGLs on a BTU basis. We noted that while the Frac Spread is a good indicator of the relative health of natural gas processing over time, it is not representative of the specific processing margin for a particular stream of input gas. That is because the Frac Spread does not take into account the quality of the gas being processed either in terms of the liquids content or the BTU content, nor does it factor in things like the operating efficiency of the plant or help determine if ethane rejection makes sense. (We’ll get to these issues in later episodes of this blog series.) These factors and others ultimately determine the quantity of NGLs that a given inlet gas stream can produce from a given gas processing plant. To incorporate these factors into gas processing margin calculations, we first have to understand how liquids content and BTU content are measured and then how to convert between gas volumes and liquid volumes, since we transform the input gas stream into both liquid and gas outputs in our processing plant. By the way, if you are a long-time reader of ours, then you will recognize this blog series as an update of our blogs a few years back titled How Rich is Rich and our Drill Down Report titled Tailgate Blues.
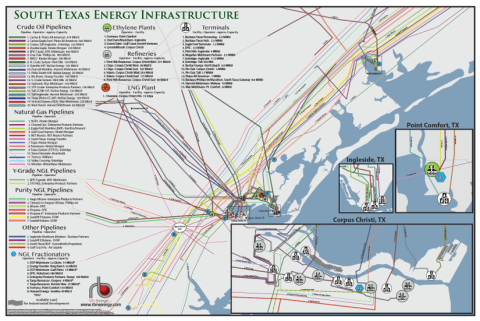
RBN Energy’s South Texas Energy Infrastructure Map brings together all the pieces of the critical and complex puzzle of the greater Corpus Christi region. Spanning from Point Comfort, TX to Corpus Christ, TX and south of the Agua Dulce natural gas hub, the map details the processing, transportation and export facilities in RBN Energy’s classic clear, concise and easy to comprehend style.
So to get everyone on the same page, we need to spend a few minutes on the basics of gas processing. Skip this section if you feel that you are up to speed on what happens in a gas processing plant.
The makeup of “raw” gas that is input into a gas processing plant (the “inlet” gas from multiple wellheads) can be simplified to three main components: methane, NGLs and everything else, a.k.a. impurities. Methane is the hydrocarbon we generally think of as natural gas (95% of the gas that moves in natural gas transmission pipelines to end-use markets is composed of methane molecules). NGLs — ethane, propane, normal butane, isobutane and natural gasoline — are the heavier hydrocarbons that come out of the wellhead mixed in with the methane. Also mixed in with the methane and NGLs are various impurities: carbon dioxide, water vapor, hydrogen sulfide (H2S), helium, nitrogen, oxygen and other undesirables. Some of these impurities are knocked out of the gas stream by treating before the gas gets to a processing plant. Other impurities are removed as part of processing within the plant.
There is no such thing as a typical gas inlet stream, but to give you some sense of the breakdown you can consider 80% methane, 15% NGLs and 5% impurities as representative of “rich” gas (explained below). Note that methane and NGLs are hydrocarbons — they burn. Impurities generally don’t burn, neither do they have value as part of the hydrocarbon stream. (A couple do have value and are processed and sold — helium for example — but they are a miniscule part of the processing economics so we are going to ignore them in this analysis).

About the song
"Good to Be Me" is a song recorded by Uncle Kracker and Kid Rock. It was released in 2010 as the second single from Kracker's album Happy Hour. The song was written by Matthew Shafer (a.k.a. Uncle Kracker), Brett James, J.T. Harding, and Robert Ritchie (a.k.a. Kid Rock). It went to #29 on Billboard’s U.S. Adult Top 40 charts and hit #28 on the U.S. Hot Country Songs chart.




